The National Institutes of Health (NIH) will implement an updated format for Data Management and Sharing Plans (DMSPs) effective May 25, 2026 (NOT-OD-26-046). While the underlying 2023 NIH Data Management and Sharing Policy remains unchanged, this new format reflects NIH’s broader effort to reduce administrative burden and improve consistency in how plans are written, reviewed, and monitored.
Understanding these updates now will help ensure a smoother transition and stronger proposals. While questions remain as NIH rolls out this format, this post outlines key implications based on what we know so far. We will continue to share updates as more details become available, including:
- whether the format will be required uniformly across all NIH Institutes and Centers or if ICs will have flexibility to customize elements, and
- how strictly NIH will enforce word limits (e.g., 200 words in Q4 and 100 words in Q6).
A More Structured, Simplified Format
One of the most significant changes is the shift from a narrative-based plan covering six elements (NOT-OD-21-014) to a structured format with Yes/No questions (Q1-3, Q5, and Q7) requiring explanations only for “No” responses or when sharing will be limited.
While this simplifies the writing process, it also shifts the focus toward clear commitments and compliance rather than descriptive planning.
Figure 1 shows a preview of the new DMS Plan (DMSP) format to be used, followed by a breakdown of how to approach each question.

Figure 1: Preview of the new DMS Plan format to be used:
Data Management and Sharing Plan Format Page (OMB Clearance Pending) (PDF, 110 KB)
Key Questions to Understand
Q1. Maximum appropriate sharing
You should generally answer “Yes,” meaning data will be shared as openly as possible and as restricted as necessary. If sharing is limited (e.g., controlled access), provide justification in Q4.
Q2: Timing of data sharing
Data must be shared by the earlier of:
- Publication, or
- End of the performance period (including no-cost extensions)
Answer “Yes” unless specific exceptions apply (e.g., SBIR/STTR or patent considerations)
- FAQ B.6: “Do SBIR/STTR projects have to share scientific data under the DMS Policy?”
- FAQ B.15: Do scientific data underlying an invention that is the subject of a patent application need to be shared? If so, when?
Q3. Data availability duration
This refers to whether data will remain available according to repository policies. In practice, this means selecting a repository that meets:
- NIH retention expectations (≥3 years)
- Institutional policies (e.g., WashU: 6 years)
- Journal requirements
Most NIH-recommended repositories meet these criteria, so “Yes” is typically appropriate.
Q4. Justification for limiting sharing of data (200 words maximum)
Use this section to explain any restrictions on sharing, including ethical, legal, or technical reasons. Refer to FAQ B.5 (What are the justifiable reasons for limiting sharing of data).
NIH considers certain reasons appropriate for limiting data sharing, such as:
- Participant privacy and confidentiality risks
- Informed consent limitations
- Legal or regulatory constraints
- Technical limitations that cannot be reasonably addressed
In contrast, the following are not considered justifiable reasons:
- The dataset is small
- The data are not expected to be widely used
- A suitable repository is not readily identified
For example, sharing may be limited when there is a high risk of participant re-identification, even after de-identification. This can occur in studies involving small sample sizes, rare diseases, or populations with distinctive characteristics, especially when combined with detailed clinical or imaging data and a geographically limited recruitment area. In such cases, investigators may determine that data cannot be shared without compromising participant privacy.
Justifications should clearly describe the specific risks and constraints that prevent data sharing.
Q5. Human participant protections
This confirms compliance with NOT-OD-22-213. A “No” response would indicate non-compliance, so investigators should familiarize themselves with NOT-OD-22-213 and plan accordingly.
Q6. Data type and repository table (100 words maximum)
This is the most important new element. Investigators must list:
- Data types, including species and modality (e.g., human scRNA sequencing data, mouse flow cytometry data, etc.)
- Associated repositories
NIH allows flexibility (“if known”), so include expected data types and refine later during RPPR if needed.

Figure 2: Question 6 of the new 2026 DMS Plan format requiring a data type and repository table
For selecting a suitable data repository for your data type, please refer to the repository selection decision tree and the resources for finding a repository.
Q7. Genomic Data Sharing (GDS) Policy compliance
This question applies to studies subject to the NIH Genomic Data Sharing (GDS) Policy (e.g., large-scale human genomic data). It asks whether data will be shared in a NIH-designated repository according to GDS timelines and whether you will meet the expectations of the Institutional Certification requirements outlined in the GDS policy (NOT-OD-14-124).
Similar to Q5, this question is intended to confirm awareness and compliance with an existing NIH GDS policy. In most cases, the appropriate response is “Yes” if your study falls under the GDS Policy. A “No” response would require justification in Q4 and may indicate non-compliance.
In limited cases, a “No” response may be appropriate, such as when:
- The informed consent does not permit broad genomic data sharing (e.g., legacy datasets)
- Legal or regulatory restrictions limit data sharing (e.g., state laws or international regulations)
- Tribal or community governance policies restrict deposition in external repositories
- There is a high risk of re-identification that cannot be adequately mitigated
- Institutional Certification requirements cannot be met due to consent or data use limitations
These scenarios should be clearly justified in Q4.
Designed for Compliance Tracking
The new format appears to be designed to support tracking during RPPR.
As shown in Figure 3, investigators will need to report:
- Whether each data type has been shared
- Reasons for delays or changes
- Updates to repositories or timelines
If significant changes occur, an updated DMSP must be submitted for NIH approval. See NOT-OD-24-176 (Updated Processes for Requesting Revisions to an Approved Data Management and Sharing (DMS) Plan) for more details.
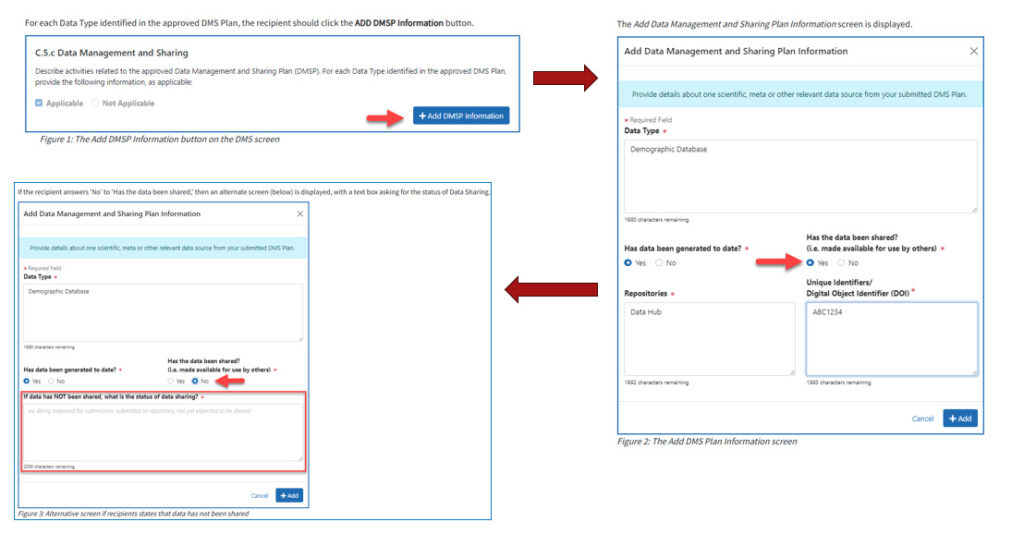
Figure 3: How to report DMS activities in RPPR
A Note of Caution: Don’t Skip the Planning Mindset
While the structured format reduces administrative burden, it may also reduce the intentional planning process that narrative plans encouraged.
The process of thinking through how data will be managed, documented, and shared across the research lifecycle is still essential. The new format does not replace the need for careful planning, it simply standardizes how that information is reported.
As data sharing becomes increasingly mandatory and scrutinized, following best practices for data management is more important than ever. Thoughtful planning helps ensure that data can be shared smoothly and in alignment with FAIR (Findable, Accessible, Interoperable, and Reusable) principles.
Support for Making Data FAIR and Reproducible
At WashU Medicine, researchers have access to Digital Commons Data@Becker, a generalist institutional data repository that supports FAIR data sharing.
In addition, the Becker Library Data Management and Sharing team provides free data curation and deposition support.
We also:
- Help identify appropriate domain-specific repositories
- Support data preparation and submission
- Provide guidance on documentation and metadata
WashU researchers also have access to protocols.io, which supports sharing reproducible, citable research methods.
To learn more, check out the “How to Make Your Protocols Complete, Interactive, and Citable” blog post and past protocols.io webinars on the Becker DMS website.
For customized guidance for your needs, please request a consultation by submitting this online form.
What This Means for Researchers
These updates reinforce that the DMSP is an operational document, not just a proposal requirement. Researchers should:
- Plan data management early
- Align plans with actual workflows
- Coordinate with institutional support teams
Start Preparing Now
Although the new format becomes effective May 25, 2026, now is the ideal time to prepare.
DMP Tool is expected to release an updated NIH DMSP template aligned with the new format, consolidating the existing NIH templates currently available in the platform. We will offer a webinar to walk you through the new template once it becomes available.
At Becker Library, we are incorporating these changes into consultations and will continue sharing example DMSPs through the ICTS WUSTL Grants Library as they become available.
If you have any questions or would like assistance, please contact the Becker Library Data Management and Sharing team at beckerdms@wustl.edu.
